The science behind Souvenaid®
Why was Souvenaid® developed?
Alzheimer’s disease damages the brain by affecting neurons (nerve cells) and the synapses, which are the connections between them. This disruption interferes with how brain cells function and communicate.
Early symptoms of Alzheimer’s disease include changes in a person’s memory, thinking and ability to interact socially. There is currently no cure for Alzheimer’s disease.1
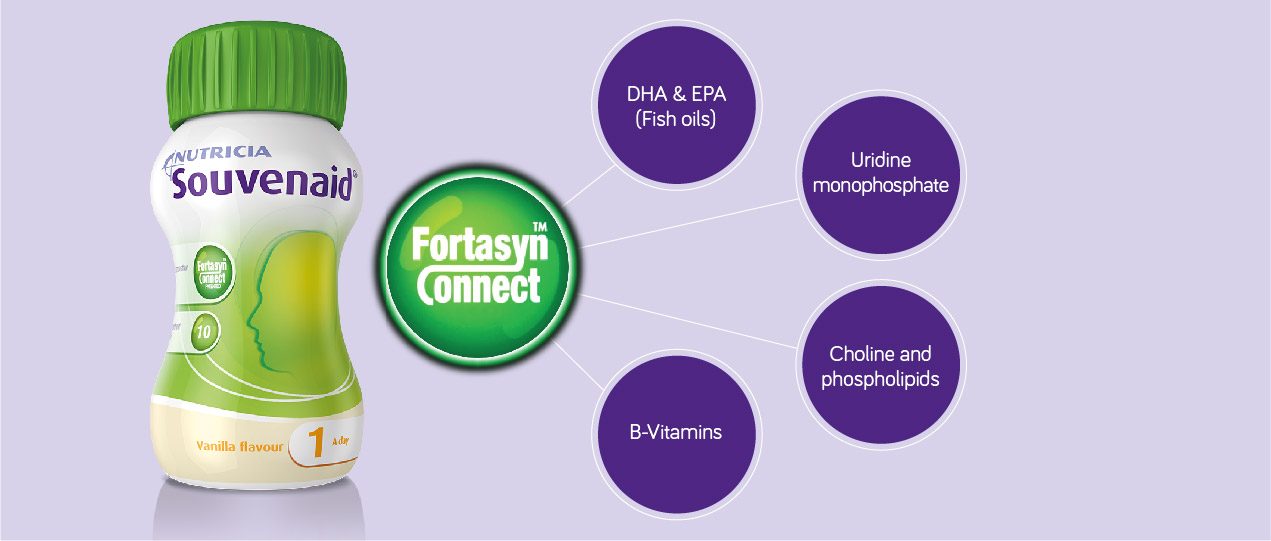
What we consume in our daily diet plays an important part in this process because the brain needs key nutrients – omega-3 fatty acids, phospholipids, choline, uridine monophosphate and B vitamins – in the right combination and at the right level to replace these synapses. 2-3
Research has shown that people with Alzheimer’s disease including mild cognitive impairment (MCI) often have low levels of some of these key nutrients including vitamins A, C, E, folate, and vitamin B12, Omega-3. 2-3
Souvenaid® in summary
Souvenaid® has been specially formulated to provide these essential nutrients at levels otherwise difficult to achieve through normal diet alone. 2-3
Souvenaid® contains a unique combination of key nutrients called Fortasyn® Connect, formulated to nutritionally support the growth of brain connections (synapses),4 and has been proven to nutritionally support memory function in the early stages of Alzheimer’s disease, including MCI. 5-7
Gerald Quigley, Community Pharmacist for over 50 years, explains Souvenaid® in this video:
Souvenaid® key memory and cognitive function results
The LipiDiDiet Clinical trial published in 2021, was an independently initiated study predominantly funded by the European Commission, which investigated the long-term use and impact of Souvenaid® on memory and cognitive function in the early stage of Alzheimer’s disease known as mild cognitive impairment.7
In this clinical trial, Souvenaid® was proven to slow memory and cognitive decline by an average of 60% in an early stage of Alzheimer’s disease known as mild cognitive impairment when taken daily for 3 years.7*
Souvenaid® nutritionally supports memory function5-7
Earlier studies completed using Souvenaid® demonstrated that Souvenaid® nutritionally supports memory function in early Alzheimer’s disease when taken daily for at least 6 months. 5-6
Souvenaid® is scientifically backed by:
- The world’s first successful long-term clinical trial in mild cognitive impairment using a nutritional intervention. 7
- 20 years of evidence-based research supported by an ongoing research program.5-8
Get professional advice
Speak to your doctor or healthcare professional about Souvenaid®. Discuss whether Souvenaid® may be a suitable inclusion in your diet to support your nutrition in the early stages of Alzheimer’s disease.
*Based on a study published in 2021. Individual results may vary.
#This product does not cure or alleviate Alzheimer’s disease. Souvenaid® is a Food for Special Medical Purposes for the dietary management of early Alzheimer’s disease. Use under medical supervision.
The information on this page is not intended to be a substitute for medical advice. Always seek medical advice from a healthcare professional.
What if I still have questions?
Take a look at our Frequently Asked Questions or get in touch with the Nutricia Careline. Our caring team of nutritionists and dietitians can answer your Souvenaid questions over phone on 1800 438 500 between 7.30am - 5pm AEST or via email using the Contact Us form.




